Types of vaccines
Vaccines: Types & Classification
Comprehensive Notes for Nursing Students
Introduction to Vaccines
Vaccines are biological preparations that provide active acquired immunity against specific infectious diseases. They are critical tools in preventive healthcare that have dramatically reduced the incidence of many potentially lethal diseases worldwide.
Key Concept:
Vaccines train the immune system to recognize and combat pathogens by introducing a harmless form of the pathogen or its components, triggering an immune response without causing disease.
Understanding the different types of vaccines, their mechanisms, and appropriate use is essential for nursing practice, especially in community health, pediatric care, and patient education.
Vaccine Classification Overview
Vaccines are classified based on their composition and mechanism of action. The main categories include:
Based on Composition
- Live-attenuated vaccines
- Inactivated (killed) vaccines
- Subunit vaccines
- Toxoid vaccines
- mRNA vaccines
- Viral vector vaccines
Based on Valency
- Monovalent - targets a single strain/serotype
- Bivalent - targets two strains/serotypes
- Trivalent - targets three strains/serotypes
- Tetravalent - targets four strains/serotypes
- Pentavalent - targets five agents/diseases
Vaccine Classification Mind Map

Figure 1: Vaccine Types and Their Mechanisms of Action
Detailed Explanation of Vaccine Types
1. Live-Attenuated Vaccines
Live-attenuated vaccines contain a weakened (attenuated) form of the pathogen that causes a disease. These vaccines closely resemble a natural infection, providing strong and long-lasting immunity, often with just one or two doses.
Important Considerations:
- Not suitable for immunocompromised patients or pregnant women
- Requires careful storage and handling (cold chain)
- May cause mild symptoms resembling the disease
- Possibility of reversion to virulence (rare)
Examples of Live-Attenuated Vaccines:
- Measles, Mumps, and Rubella (MMR)
- Varicella (Chickenpox)
- Rotavirus
- Yellow Fever
- Oral Polio Vaccine (OPV)
- BCG (Tuberculosis)
Mechanism of Action

Figure 2: How Live-Attenuated Vaccines Work
2. Inactivated (Killed) Vaccines
Inactivated vaccines contain pathogens that have been killed through physical or chemical processes. These vaccines cannot cause disease because the pathogen is dead, making them safer for immunocompromised individuals.
Key Characteristics:
- Cannot replicate in the body
- Usually requires multiple doses for primary series
- Often requires adjuvants to enhance immune response
- Typically needs booster doses to maintain immunity
- More stable than live vaccines (may not require constant refrigeration)
Examples of Inactivated Vaccines:
- Inactivated Polio Vaccine (IPV)
- Hepatitis A
- Rabies
- Some Influenza vaccines (flu shot)
- Whole-cell Pertussis (in DTP vaccine)
- Cholera
Clinical Note:
Inactivated vaccines typically induce a predominantly humoral (antibody) immune response and may not trigger strong cellular immunity. This is why multiple doses are often required to build and maintain adequate protection.
3. Subunit, Recombinant, Polysaccharide, and Conjugate Vaccines
Instead of the entire pathogen, these vaccines include only specific parts (subunits) that best stimulate the immune system. They can be created using various approaches:
| Type | Description | Examples |
|---|---|---|
| Subunit | Contains purified fragments of the pathogen (proteins, polysaccharides) | Hepatitis B, Acellular Pertussis (in DTaP/Tdap) |
| Recombinant | Uses genetic engineering to produce specific antigens | HPV, Hepatitis B, Shingles (Shingrix) |
| Polysaccharide | Contains sugar molecules from bacterial capsules | Pneumococcal (PPSV23), Meningococcal, Typhoid Vi |
| Conjugate | Polysaccharides linked to carrier proteins to enhance immunogenicity | Hib, Pneumococcal (PCV13), Meningococcal conjugate |
Clinical Significance:
Conjugate vaccines represent a significant advancement over plain polysaccharide vaccines. By attaching the polysaccharide to a protein carrier, they convert a T-independent immune response to a T-dependent one, improving immunogenicity in young children and producing immune memory.
Advantages:
- Highly specific immune response
- Cannot cause disease (very safe profile)
- Suitable for immunocompromised individuals
- Often easier to produce and store
Limitations:
- May require adjuvants to boost immune response
- Multiple doses typically needed
- May induce shorter duration of immunity
- More expensive to develop and produce
4. Toxoid Vaccines
Toxoid vaccines use inactivated bacterial toxins (toxoids) to immunize against diseases where the primary cause of illness is the toxin produced by the bacteria, rather than the bacteria itself.
Mechanism of Action:
These vaccines work by chemically inactivating the toxin (usually with formaldehyde) to eliminate its harmful effects while maintaining its ability to trigger an immune response. When the immune system encounters the actual toxin later, it recognizes it and neutralizes it before damage occurs.
Key Examples:
- Tetanus toxoid - Found in DT, DTaP, Tdap, and Td vaccines
- Diphtheria toxoid - Found in DT, DTaP, Tdap, and Td vaccines
Clinical Application
Tetanus toxoid vaccination is crucial for wound management. If a patient has a tetanus-prone wound and:
- Has had <3 doses or unknown history: Give TIG + Td/Tdap
- Has had ≥3 doses but last dose >5 years ago (for dirty wounds) or >10 years (for clean wounds): Booster dose needed
TIG = Tetanus Immune Globulin
5. mRNA Vaccines
mRNA vaccines represent a revolutionary approach in vaccine technology. While the platform gained worldwide attention with COVID-19 vaccines, the technology had been under development for decades before its widespread implementation.
Mechanism of Action:
mRNA vaccines deliver genetic instructions for cells to produce a specific protein (usually a viral surface protein). The cells then display this protein, triggering an immune response. The mRNA never enters the cell nucleus or alters DNA.
Key Advantages:
- Rapid development and manufacturing
- No live components - cannot cause infection
- No integration with host DNA
- Strong immune response (both humoral and cellular)
- Can be adapted quickly for new variants
mRNA Vaccine Process

Figure 3: How mRNA Vaccines Generate Immunity
Storage Considerations:
mRNA is inherently unstable, requiring special storage conditions. The first generation of COVID-19 mRNA vaccines needed ultra-cold storage (-70°C for Pfizer initially, -20°C for Moderna). Newer formulations have improved stability, but cold chain management remains crucial.
Current Examples:
- COVID-19 vaccines (Pfizer-BioNTech, Moderna)
- Research ongoing for influenza, CMV, Zika, and cancer vaccines
6. Viral Vector Vaccines
Viral vector vaccines use a modified version of a different virus (the vector) to deliver important genetic material from the target pathogen to cells. The vector virus is altered so it cannot cause disease but can efficiently transport the genetic instructions to produce antigens.
How They Work:
- A harmless viral vector (often adenovirus) is modified to carry genetic material from the target pathogen
- The vector enters cells and delivers genetic instructions
- Cells produce the target antigen (e.g., spike protein for COVID-19)
- The immune system recognizes the antigen as foreign
- Antibodies and T-cells develop against the antigen
Examples:
- COVID-19: Oxford/AstraZeneca, Johnson & Johnson (Janssen), Sputnik V
- Ebola: Ervebo
- In development for HIV, Zika, and other diseases
Advantages & Limitations
Advantages:
- Single dose may be sufficient
- Strong cellular immune response
- No adjuvant needed
- More stable than mRNA (standard refrigeration)
- Technology adaptable to multiple diseases
Limitations:
- Pre-existing immunity to vector may reduce effectiveness
- Complex manufacturing process
- Some associated with rare adverse events (e.g., thrombosis with thrombocytopenia)
- Cannot be used repeatedly with same vector
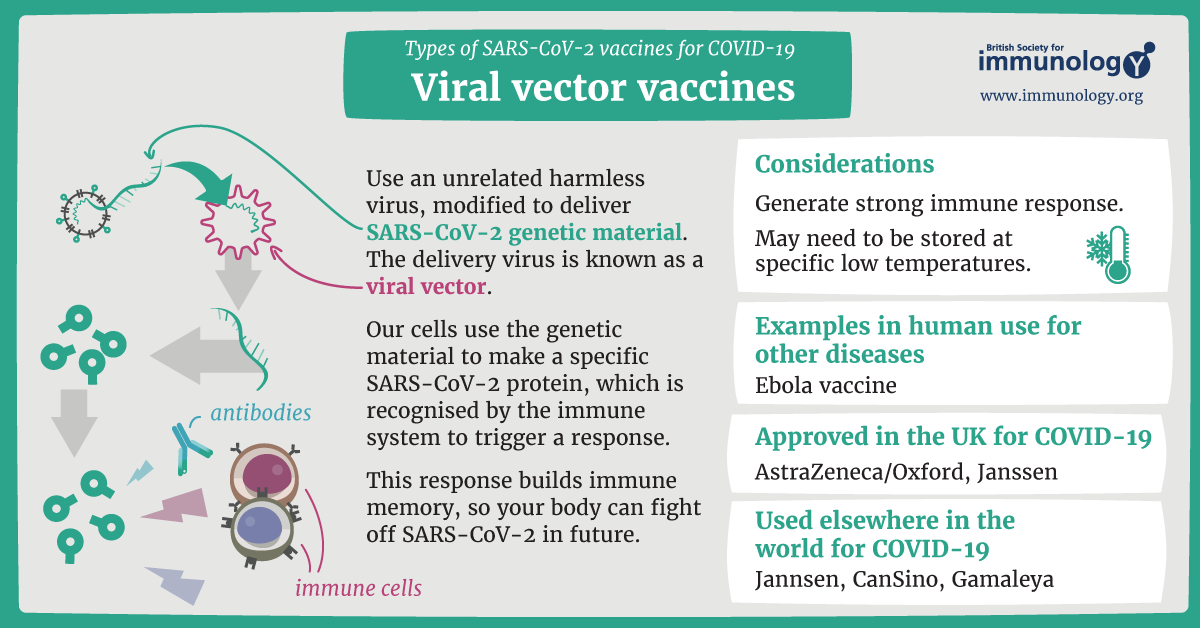
Figure 4: Mechanism of Action of Viral Vector Vaccines
Helpful Mnemonics for Vaccine Classification
LIVE MTR
Remember common live-attenuated vaccines:
- L - Live attenuated influenza (nasal)
- I - Intranasal influenza
- V - Varicella (chickenpox)
- E - Everything yellow fever
- M - Measles
- T - TB (BCG)
- R - Rotavirus, Rubella
KIPER
Remember common inactivated (killed) vaccines:
- K - Killed organisms
- I - Influenza (injectable)
- P - Polio (IPV)
- E - Encephalitis (Japanese)
- R - Rabies
TORCH Vaccines
A guide to remember vaccines related to TORCH infections:
- T - Toxoplasmosis (no vaccine available)
- O - Other infections (Syphilis, HIV, etc. - limited vaccines)
- R - Rubella (MMR vaccine)
- C - Cytomegalovirus (no approved vaccine yet)
- H - Herpes simplex (no approved vaccine yet)
Note: While not all TORCH infections have vaccines, this mnemonic helps remember which maternal infections are vaccine-preventable (primarily Rubella).
Vaccine Types: Comprehensive Comparison
| Vaccine Type | Composition | Immune Response | Advantages | Limitations | Dose Requirements |
|---|---|---|---|---|---|
| Live-Attenuated | Weakened pathogen | Strong humoral and cellular immunity | Long-lasting, often lifetime immunity | Contraindicated in immunocompromised | Often single or few doses |
| Inactivated | Killed pathogen | Primarily humoral immunity | Safe for immunocompromised | Requires multiple doses | Multiple doses + boosters |
| Subunit | Specific pathogen fragments | Focused immune response | Very safe, minimal side effects | Less immunogenic | Multiple doses + boosters |
| Toxoid | Inactivated bacterial toxins | Antitoxin antibodies | Prevents toxin-mediated diseases | No protection against infection | Multiple doses + boosters |
| Conjugate | Polysaccharide linked to protein | Enhanced T-cell dependent response | Effective in infants | Complex manufacturing | Multiple doses based on age |
| mRNA | Genetic instructions for antigen | Strong humoral and cellular immunity | Rapid development, highly specific | Storage challenges | Typically 2 doses |
| Viral Vector | Harmless virus carrying target genes | Strong cellular immunity | Durable immune response | Pre-existing vector immunity | Usually 1-2 doses |
Table 1: Comprehensive comparison of different vaccine types, their properties, and clinical considerations
Key Points Summary
- Vaccines are classified by their composition, preparation method, and mechanism of action
- Live-attenuated vaccines provide strong, long-lasting immunity but have contraindications for certain populations
- Inactivated vaccines are safer but typically require multiple doses and boosters
- Subunit, recombinant, and toxoid vaccines focus on specific components of pathogens
- Conjugate vaccines significantly improved protection for infants against encapsulated bacteria
- mRNA and viral vector vaccines represent newer technologies with unique advantages
- Vaccine selection depends on pathogen characteristics, target population, and practical considerations like storage and cost
- Understanding vaccine types helps nurses provide appropriate patient education and assess contraindications
References
- Centers for Disease Control and Prevention. (2022). Vaccine Types. https://www.cdc.gov/vaccines/hcp/admin/storage-handling.html
- U.S. Department of Health & Human Services. (2022). Vaccine Types. https://www.hhs.gov/immunization/basics/types/index.html
- World Health Organization. (2023). Vaccines and immunization: What is vaccination? https://www.who.int/news-room/questions-and-answers/item/vaccines-and-immunization-what-is-vaccination
- National Institute of Allergy and Infectious Diseases. (2019). Vaccine Types. https://www.niaid.nih.gov/research/vaccine-types
- British Society for Immunology. (2023). Types of vaccines for COVID-19. https://www.immunology.org/public-information/vaccine-resources/covid-19/covid-19-vaccine-infographics/types-covid19-vaccines
Comments
Post a Comment